 16,26–29 This deficit largely stems from the inability to grow high-quality single crystals of ThO 2 with confirmed crystal orientation, purities, and stoichiometries. 25 While important inroads have been made in understanding the role of small-scale defects on thermal transport in UO 2, a similar level of experimentally validated understanding for ThO 2 is lacking. One notable recent example is the work of Khafizov and co-workers who used a combination of microscale thermal transport measurements and XRD to infer features of nanoscale defect evolution in ion-irradiated UO 2. For both UO 2 and CeO 2, combinations of more easily implemented benchtop methods including XRD, 22 Raman scattering, 23 and other optical techniques 24 have been used to characterize defect populations in these oxides. 19 Synchrotron-based methods including X-ray diffraction (XRD) 20 and absorption methods 21 have also been used to collect detailed local defect structures in fluorite oxides. Positron annihilation spectroscopy (PAS) has been used to characterize defects on the smallest scales, however, PAS is only sensitive to vacancy-type defects and cluster type discrimination is difficult. 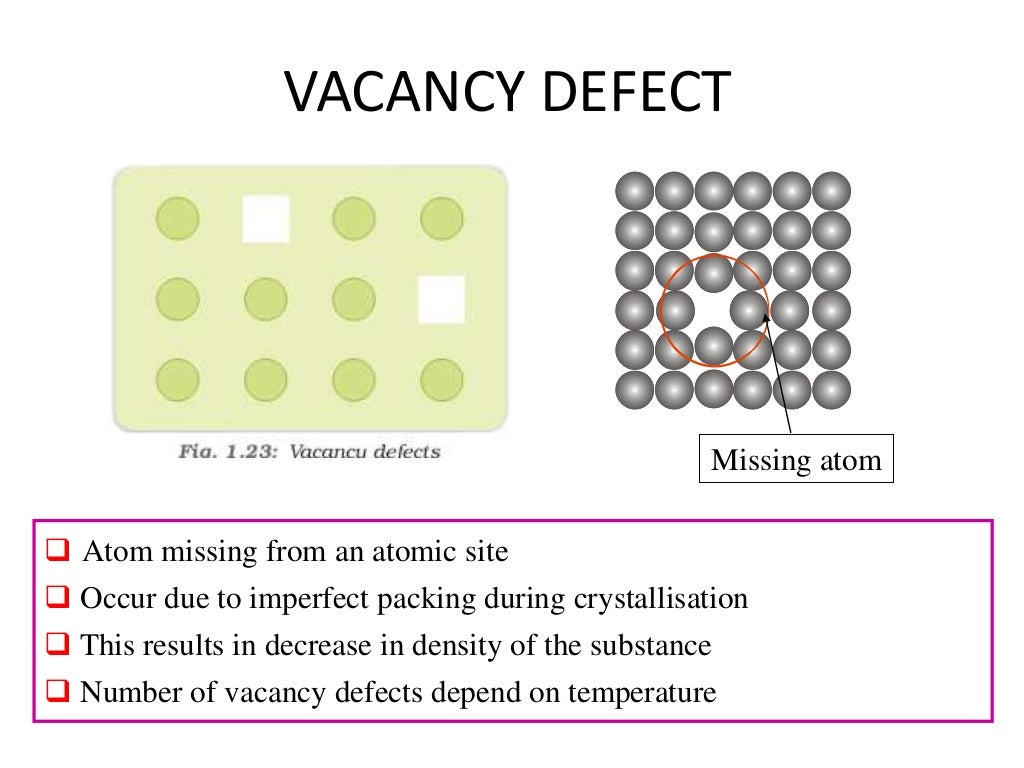 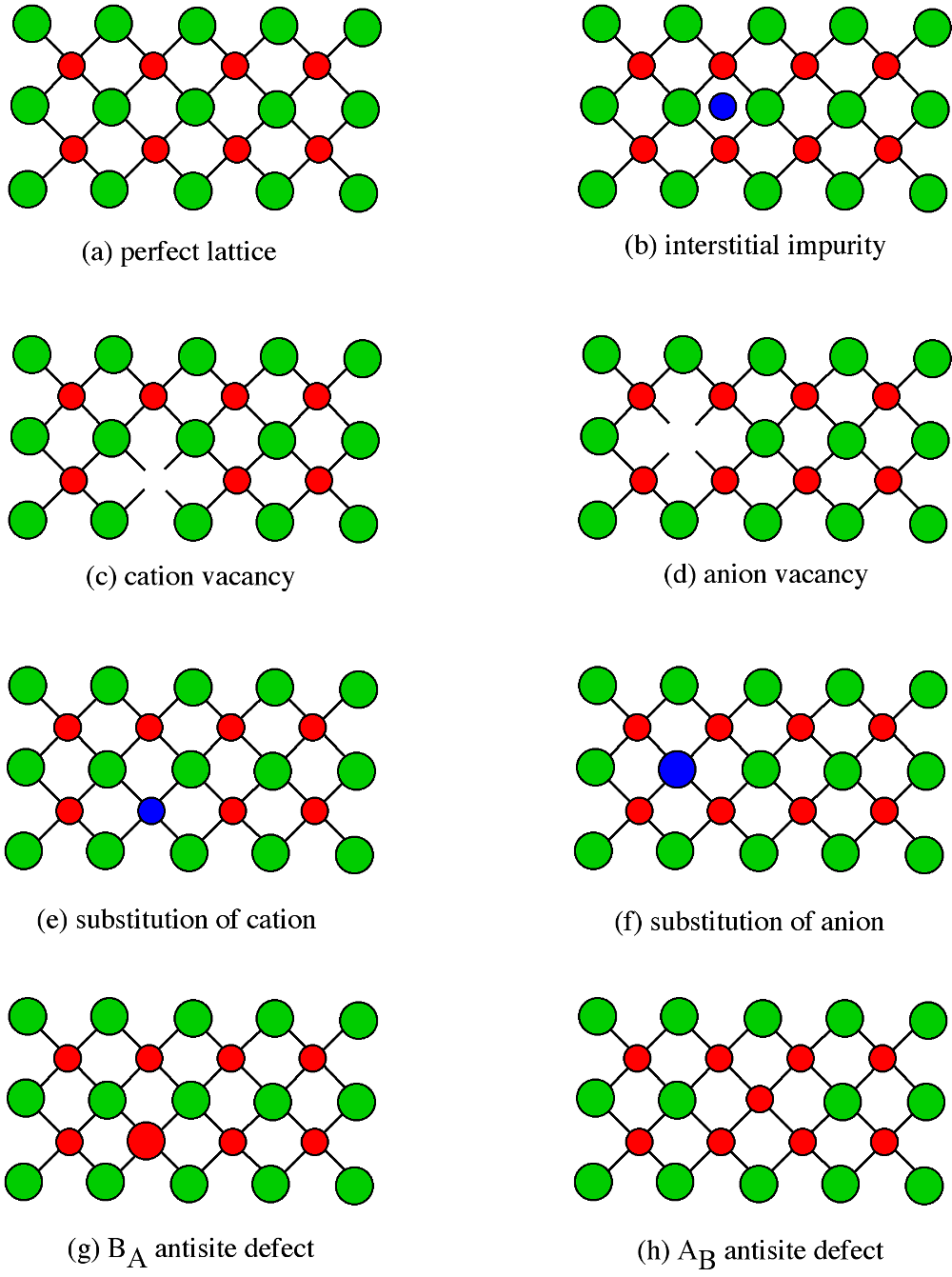
Therefore, indirect methods must be used to characterize the presence of these sub-resolution defects. 18 Characterizing both the size and concentration of these types of defects is challenging as statistically significant populations of defects cannot be imaged directly using ultrahigh resolution microscopy. 15–18 However, it has been shown in actinide and lanthanide oxides that the largest effects on thermal transport are often caused by the smallest-scale lattice defects at the earliest stages of damage accumulation. These nanoscale to microscale features have been revealed to drastically reduce thermal conductivity. Potential operating environments for these materials include high radiation fields and extreme temperatures that promote or directly generate lattice defects in otherwise perfect fluorite structures. 13,14 For many of these applications, thermal transport is a key property that determines the suitability of a particular oxide for a particular use, controlling, for example, the peak center line temperature in nuclear fuels and heat dissipation ability in large bandgap electronics. 12 Exhibiting similar behavior in many respects, ThO 2 has several key distinctions from other heavy metal fluorite oxides that make it attractive for the above mentioned applications including a fixed tetravalent cation oxidation state, extremely high melting temperature, and large electronic bandgap. 5–8 In contrast, ThO 2 has been less widely investigated to date despite potential applications as a fertile fuel in advanced, proliferation-resistant nuclear reactors 9–11 and as a high reflectivity material for extreme ultraviolet optics. 2–4 Given these technological implications, the thermophysical properties and performance characteristics of UO 2 and CeO 2 have been the subject of detailed study for decades. 1 CeO 2 is utilized in electrochemical applications as a catalysis material due to its ability to store and transport oxygen and as a solid oxide fuel cell material. 
UO 2 forms the basis for the large majority of commercial nuclear fuels worldwide. These methods also provide a promising pathway for the quantification of the smallest-scale defects that cannot be captured using traditional microscopy techniques and play an outsized role in degrading thermal performance.Īctinide and lanthanide fluorite oxides, ThO 2, UO 2, and CeO 2, form an important family of high temperature ceramics for a variety of energy applications. Together, this combination of methods provides important initial insight into defect formation, recombination, and clustering in thorium dioxide and the effect of those defects on thermal transport. Defected regions are investigated using spatial domain thermoreflectance to quantify the change in thermal conductivity as a function of ion fluence as well as transmission electron microscopy and Raman spectroscopy to interrogate the structure of the generated defects. In this work, a series of high-quality thorium dioxide single crystals are exposed to 2 MeV proton irradiation at room temperature and 600 ☌ to create microscale regions with varying densities and types of point and extended defects. An understanding of the effect of nanoscale lattice defects on thermal transport in this material is currently unavailable due to the lack of a single crystal material from which unit processes may be investigated. Thermal transport is a key performance metric for thorium dioxide in many applications where defect-generating radiation fields are present. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
